Purpose:
The purpose of this lab is to figure out which native plants have active ingredients that inhibit the growth of bacteria.
The purpose of this lab is to figure out which native plants have active ingredients that inhibit the growth of bacteria.
|
Materials:
Plant specimen (persimmon leaf) 10 mL syringe test tubes Methanol 1 mL pipet Ampicillin Inoculating Loop 60x15 Petri dish |
E. Coli JM109 (stock plate) 10 mL pipet 100 mL beakers 5mm filter paper 250 mL media bottle LB Agar LB broth base Plastic funnels |
Procedure:
Part 1:
1. Prepare an LB broth for the E. coli. Wait 24 hours and then add a colony of the E. coli to the broth.
2. Get six petri dishes. Draw a "+" symbol on each plate and then divide the dish into 4 quadrants. Label the quadrants 1-4 and put your initials and the date on the plate as well.
3. Liquefy LB Agar by heating it in the microwave. Pour approximately 20 ml of the agar into each petri dish. Let the agar solidify for 24 hours.
Part 2:
1. Grind up your plant specimen in deionized water. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 ml of this mixture and put it in a labeled test tube.
2. Grind up your plant specimen in methanol. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 ml of this mixture and put it in a labeled test tube. Then place the test tube in a 65 degree Celsius heat block with the cap open for 24 hours. After, put 10 ml of deionized water into the test tube and vortex it.
3. Drop 4 filter paper disks into each extract test tube (methanol and water extract tubes).
4. Prepare 2 negative control disks of methanol and distilled water.
5. Prepare 2 positive control disks of ampicillin.
6. Close tubes and store at 4 degrees Celsius.
Part 3:
1. Using a sterile 1 ml pipet, transfer 1 ml of E. coli to the petri dish and spread it over the 4 quadrants. Spread it with the glass spreader after sterilizing it with alcohol and fire. Then cover the petri dish and let it soak for 15 minutes.
2. Use forceps, sterilized with alcohol and fire, to place the discs on the plate in the appropriate place. One methanol extract disc in each MeoH quadrant, one water extract disc in each water quadrant, one ampicillin disc on the +, and one water disc on the -.
3. Incubate at 37 degrees Celsius for 24 hours.
Part 4:
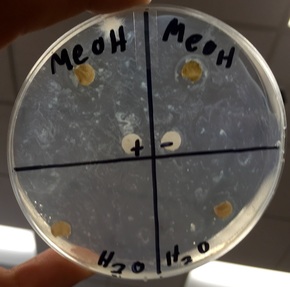
1. Observe the petri dish. If their is bacteria growing against the disk then it came out negative. If there is a ring without bacteria around the disk then it is a positive sample.
Part 1:
1. Prepare an LB broth for the E. coli. Wait 24 hours and then add a colony of the E. coli to the broth.
2. Get six petri dishes. Draw a "+" symbol on each plate and then divide the dish into 4 quadrants. Label the quadrants 1-4 and put your initials and the date on the plate as well.
3. Liquefy LB Agar by heating it in the microwave. Pour approximately 20 ml of the agar into each petri dish. Let the agar solidify for 24 hours.
Part 2:
1. Grind up your plant specimen in deionized water. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 ml of this mixture and put it in a labeled test tube.
2. Grind up your plant specimen in methanol. Filter it through filter paper. Then filter the mixture again through a syringe filter. Collect 1 ml of this mixture and put it in a labeled test tube. Then place the test tube in a 65 degree Celsius heat block with the cap open for 24 hours. After, put 10 ml of deionized water into the test tube and vortex it.
3. Drop 4 filter paper disks into each extract test tube (methanol and water extract tubes).
4. Prepare 2 negative control disks of methanol and distilled water.
5. Prepare 2 positive control disks of ampicillin.
6. Close tubes and store at 4 degrees Celsius.
Part 3:
1. Using a sterile 1 ml pipet, transfer 1 ml of E. coli to the petri dish and spread it over the 4 quadrants. Spread it with the glass spreader after sterilizing it with alcohol and fire. Then cover the petri dish and let it soak for 15 minutes.
2. Use forceps, sterilized with alcohol and fire, to place the discs on the plate in the appropriate place. One methanol extract disc in each MeoH quadrant, one water extract disc in each water quadrant, one ampicillin disc on the +, and one water disc on the -.
3. Incubate at 37 degrees Celsius for 24 hours.
Part 4:
1. Observe the petri dish. If their is bacteria growing against the disk then it came out negative. If there is a ring without bacteria around the disk then it is a positive sample.
Lab Questions:
1. When an extract gives you a negative result in the antimicrobial assay, it doesn't necessarily mean that the extract is not an antimicrobial agent. This is because we only have results of it not working on this string of E. coli, so it could potentially still work on another string of E. coli.
2. Some of the methanol extractions might smell like alcohol because we used alcohol to sterilize the tools and may not have burned all the alcohol off. This is a problem because alcohol kills bacteria causing faulty data in the end.
3. Since each extract may have multiple compounds in it, they need to be separated to find which ones are causing the antimicrobial actions. In order to find this, we would use chromatography to separate the individual molecules.
1. When an extract gives you a negative result in the antimicrobial assay, it doesn't necessarily mean that the extract is not an antimicrobial agent. This is because we only have results of it not working on this string of E. coli, so it could potentially still work on another string of E. coli.
2. Some of the methanol extractions might smell like alcohol because we used alcohol to sterilize the tools and may not have burned all the alcohol off. This is a problem because alcohol kills bacteria causing faulty data in the end.
3. Since each extract may have multiple compounds in it, they need to be separated to find which ones are causing the antimicrobial actions. In order to find this, we would use chromatography to separate the individual molecules.
Conclusion:
The only extract that gave a positive result was the methanol extract, but this may not be certain since only one of the two methanol samples showed up strongly positive. The controls did not work as expected because both of them showed up as being negative and one of them should have been positive. We could have gotten this false result because our mixtures may not have been exact or had the right ingredients. Another problem that could have happened was alcohol interference on the sterilized tools. We could improve this experiment by redoing our positive and negative controls to make sure they were right and making sure all the alcohol was burned off of sterile equipment. The next step would be to retest the methanol extract to see for sure if it is positive. If it is still positive, then we would use chromatography to isolate the part of the extract that causes it to be antimicrobial.
The only extract that gave a positive result was the methanol extract, but this may not be certain since only one of the two methanol samples showed up strongly positive. The controls did not work as expected because both of them showed up as being negative and one of them should have been positive. We could have gotten this false result because our mixtures may not have been exact or had the right ingredients. Another problem that could have happened was alcohol interference on the sterilized tools. We could improve this experiment by redoing our positive and negative controls to make sure they were right and making sure all the alcohol was burned off of sterile equipment. The next step would be to retest the methanol extract to see for sure if it is positive. If it is still positive, then we would use chromatography to isolate the part of the extract that causes it to be antimicrobial.